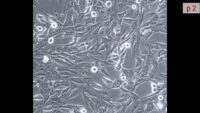
Welcome to our complete package of video tutorials on cell culture technique. All commonly used procedures in the cell culture lab are described and discussed here.
Content
We adhere to the rules of Good Cell Culture Practice and show how you can easily implement them in your lab.
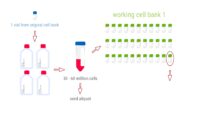
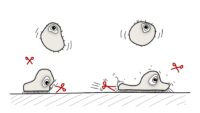
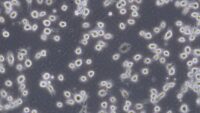
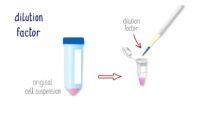
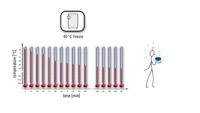
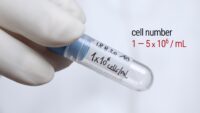
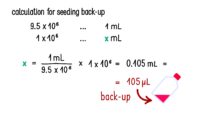
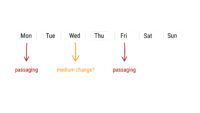
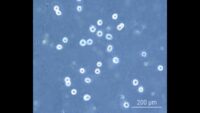
Topics range from getting ready for the cell culture lab to cells, cell handling, cell culture medium, cell morphology, cell confluence, cell types and cell line misidentification. We also discuss cell passaging, counting and freezing cells and talk about contamination in the cell culture lab.
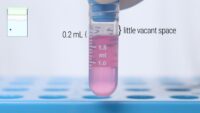
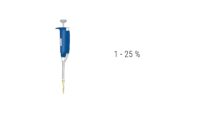
Since the vessels and equipment in the cell culture lab have a direct impact on the cells and the data we generate, a whole section is dedicated to vessels, flasks and plates. We also talk a lot about correct pipetting technique.
The tutorials include common mistakes, common misconceptions, food for thought, but also good habits, opinions, tips and hacks as well as dos and don’ts.
Who will benefit
The tutorials can be watched in class, after class or before starting a practical course, or before starting a bachelor, a master or a PhD programme. They can be watched before starting to work in a cell culture lab, or before switching to a different workplace that happens to be a cell culture lab.
We hope the videos encourage you to discuss why things are done a certain way in the cell culture lab, and where things can still be improved. Regarding the order of the videos, we recommend a certain order, but, of course, you are free to choose a different order if that makes more sense to you and your situation.